Archives of Otolaryngology and Rhinology
Morphology of the inferior laryngeal nerve (recurrent nerve) in the anterior cervical region: A report of 30 thyroidectomies
1ENT and Cervico-Facial Surgery Department, Idrissa Pouye General Hospital, Dakar, Senegal
2Laboratory of Anatomy and Organogenesis, Cheikh Anta Diop University, Dakar, Senegal
3ENT and Cervico-Facial Surgery Department, Diamniadio Children's Hospital, Dakar, Senegal
Author and article information
Cite this as
Diouf MS, Ndiaye M, Ndiaga Ndoye JM, Gaye M, Ndiaye M, et al. (2023) Morphology of the inferior laryngeal nerve (recurrent nerve) in the anterior cervical region: A report of 30 thyroidectomies. Arch Otolaryngol Rhinold. 2023; 9(2): 6-12. Available from: 10.17352/2455-1759.000151
Copyright License
© 2023 Diouf MS, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Objective: To study the morphological peculiarities of the left and right Inferior Laryngeal Nerves (ILN) in the anterior cervical region and their variations induced by thyroid pathology.
Materials and methods: The study concerned 30 patients who underwent thyroidectomy at the ENT and cervico-facial surgery department of Idrissa POUYE general hospital (HOGIP) from June 1st, 2018 to June 30, 2019. Age, sex, morphotype, operative indication, type of thyroidectomy, route, morphometry, and mode of termination of the ILN were studied.
Results: The mean age was 42 years. There were 29 women and one man. Eleven patients (36.7%) were brevilinear, 8 patients (26.6%) were longilinear and 11 patients (36.7%) were of intermediate morphotype. Multinodular goiter was the indication for surgery in 18 cases. A total of 53 ILNs were studied (29 left and 24 right). Path variations concerned 8 left recurrent nerves (28%). The mean diameter of the ILN was 2mm. Longilinear patients and those with intermediate morphotypes had the longest recurrent nerves, while brevilinear patients had the thickest nerves. On the right, the ILN was bifid in 10 cases (41.6%), trifurcated in 3 cases (12.5%), and fan-shaped in 1 case (4.1%). On the left, it was bifid in 23 cases (79.3%) and trifurcated in 2 cases (6.8%).
Conclusion: The caliber of the recurrent nerve is relatively large, and can sometimes be spindly, especially on the right. Morphotype may predict ILN dimensions. Extra-laryngeal division of the ILN is a common situation.
Historical
The recurrent nerve was discovered in the 2nd century by Galen, who described a nerve coming from the brain on either side of the neck, descending to the heart, then ascending to the larynx and causing the vocal cords to open [7]. He named it the recurrent nerve and also discovered that it was a branch of the vagus nerve since sectioning the latter in pigs caused aphonia. Galen thus confirmed that the ILN was the motor nerve of the larynx, discovering that its interruption caused severe impairment of phonation [19,20]. This discovery marked a turning point in history, proving that the voice generated by the larynx was controlled by the brain and not by the heart, as was believed at the time.
In 1898 Theodor Kocher's meticulous technique resulted in an incidence of recurrent nerve injury similar to that of surgeons today [20]. In 1971, Blondeau and his team demonstrated that exposure to the ILN reduced the frequency of post-thyroidectomy recurrent paralysis [21].
Materials and methods
A descriptive, cross-sectional observational study was conducted in the ENT and cervico-facial surgery department of the HOGIP from June 1st, 2018 to June 30th, 2019, for a period of 12 months. Included in the study was any patient who presented a multinodular goiter, a thyroid nodule, or Graves’ disease and had to benefit from a thyroidectomy. All patients had to be operated on by the investigator himself, to avoid bias due to inter-individual variability. Anthropometric characteristics (age, sex, morphotype), operative indication, and type of thyroidectomy were studied.
As regards the ILN, we studied its course, length, caliber, the existence of extra-laryngeal nerve division branches, and the level of laryngeal penetration in relation to the Small Horn of the Thyroid Cartilage (SHTC). All patients underwent preoperative and postoperative indirect laryngoscopy or nasofibroscopy to rule out recurrent nerve paralysis.
The following measuring devices were used: a protractor to measure the infrasternal angle, a camera for archiving purposes, and a ruler to measure the length and estimate the diameter of the recurrent nerve using a suture thread brought back to the ruler. The length of the ILN was measured on the right and left sides between the upper edge of the clavicle and the cricoid-tracheal angle, and expressed in centimetres (cm). The diameter was measured at the intersection with the Inferior Thyroid Artery (ITA) and expressed in millimeters (mm).
Ethical Consideration
Informed consent was obtained from all patients before the operation and the study was conducted according to the principles of the Declaration of Helsinki.
Results
A total of 30 patients were selected. The mean age was 42 years, with extremes of 23 and 74 years. Twenty-nine patients (96.7%) were female and one patient (3.3%) was male. The sex ratio was 0.03. With regard to morphotype, eleven patients (36.7%) were brevilinear, 8 patients (26.6%) were longilinear and 11 patients (36.7%) were of intermediate morphotype (Table 1). All patients underwent surgery for euthyroidism under general anesthesia with orotracheal intubation. Multinodular goiter was the operative indication in 18 cases. Concerning the surgical procedure, 23 patients (77%) had undergone total thyroidectomy, left loboisthmectomy was performed in 6 patients (20%), and 1 patient (3%) had undergone right loboisthmectomy. A total of 53 ILNs were studied. These included 29 left ILNs (23 total thyroidectomies + 6 left loboisthmectomies) and 24 right ILNs (23 total thyroidectomies + 1 right loboisthmectomy).
Route
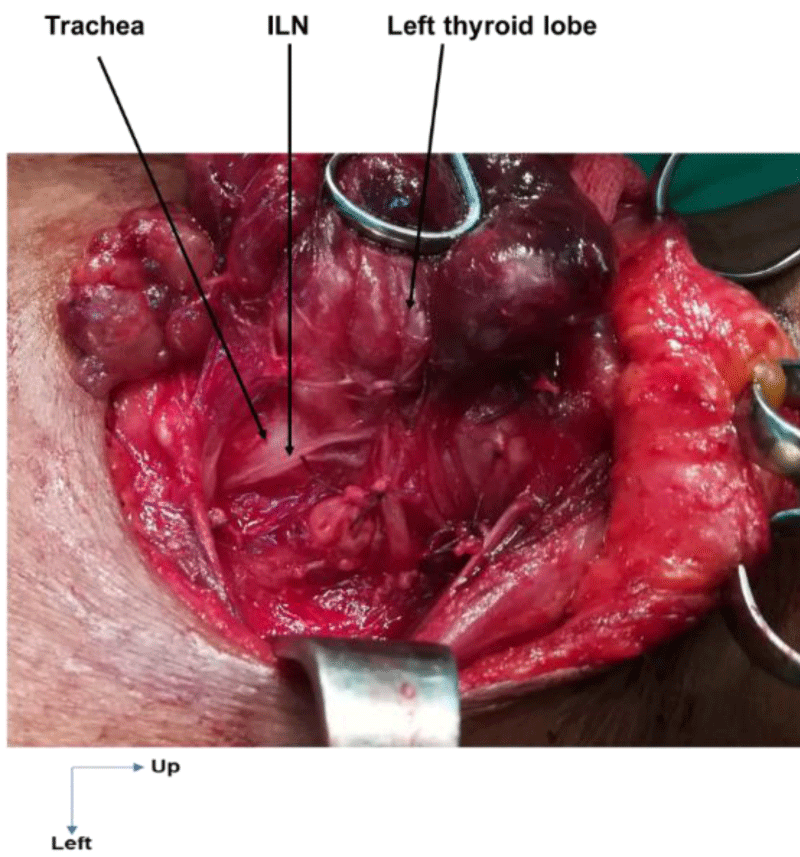
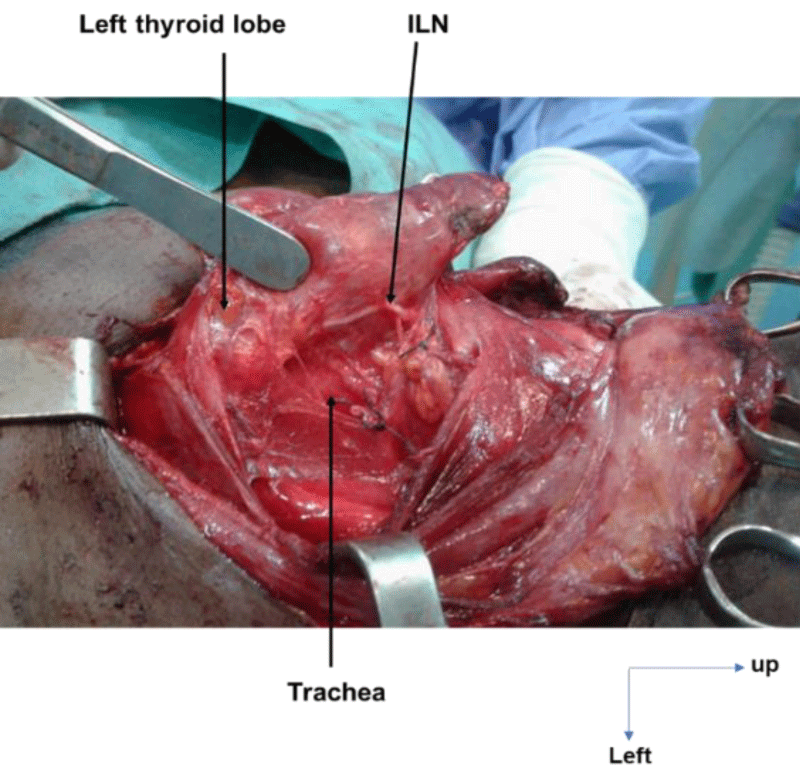
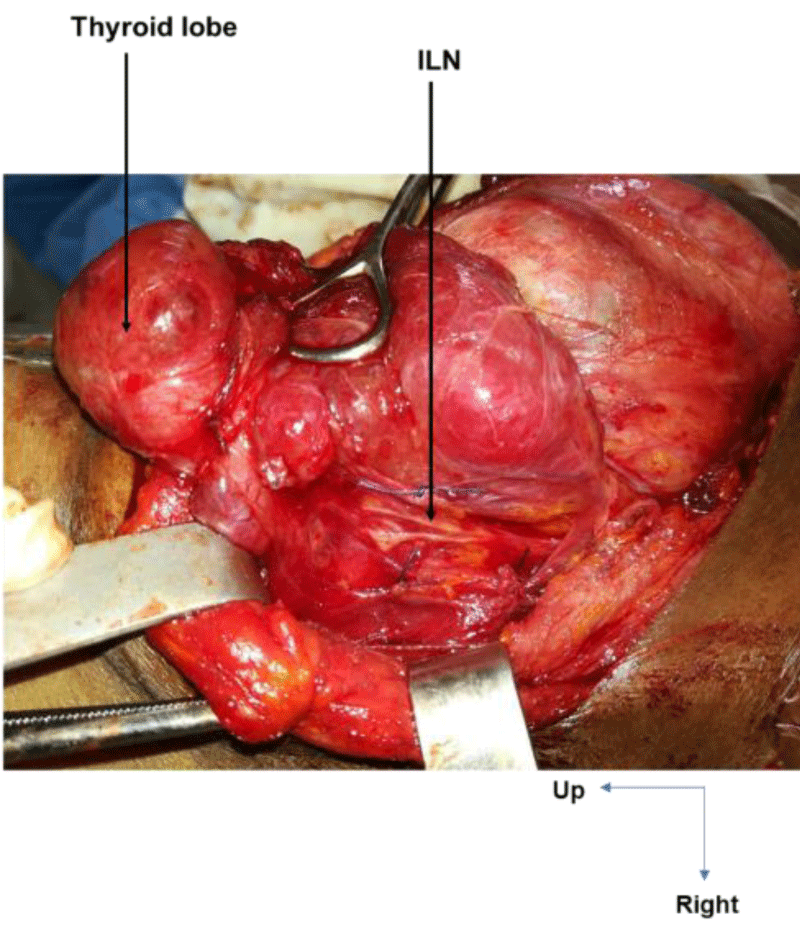
On the right, in 24 cases (100%), the ILN ascended to the cricotracheal angle, lateral to the trachea. This path was sinuous in 3 cases (12.5%) and rectilinear in 21 cases (87.5%). On the left, the ILN was located in the tracheoesophageal angle in 21 cases (72.4%), on the anterior surface of the trachea in 6 cases (20.7%, Figure 1) and on the posterior surface of the left lobe of the thyroid gland in 2 cases (6.9%, Figure 2). This path was sinuous in 5 cases (17.3%) and rectilinear in 24 cases (82.7%).
Dimensions
The mean ILN diameter was 2.3mm on the right and 2.5mm on the left, with extremes of 1mm and 5mm on the right and 1mm and 6mm on the left. Nine patients (17%) on the right and 2 patients (4%) on the left had a small nerve diameter of 1.5mm or less. Recurrent nerve diameter was greater than or equal to 3mm in 8 patients (15%) on the right and 9 patients (17%) on the left.
Length
On the right, the mean length of the recurrent nerve was 5.3 cm, with extremes of 3 cm and 11.9 cm. On the left, the average was 7.2 cm, with extremes of 3 cm and 13 cm.
In the longilinears patients, the mean caliber of the recurrent nerve was 2.3 mm on the right, with extremes of 1.5 mm and 4 mm, and 2.8 mm on the left, with extremes of 2 mm and 4 mm. The mean length of the ILN was 5.3 cm on the right and 7.6 cm on the left, with extremes of 3.5 cm and 10.8 cm on the right and 4 cm and 13 cm on the left.
In patients with an intermediate morphotype, the mean caliber of the recurrent nerve was 2.15 mm on the right with extremes of 1.5 mm and 3 mm and 2.3 mm on the left with extremes of 1.5 mm and 4 mm. The mean length of the ILN was 5 cm on the right and 7.1 cm on the left, with extremes of 3 cm and 11.9 cm on the right and 3.5 cm and 13 cm on the left.
In brevilinear patients, the mean caliber of the recurrent nerve was 2.3 mm on the right, with extremes of 1 mm and 5 mm, and 2.5 mm on the left, with extremes of 1 mm and 6 mm. The mean length of the ILN was 4.6 cm on the right and 7 cm on the left, with extremes of 3 cm and 7.5 cm on the right and 3 cm and 8.1 cm on the left.
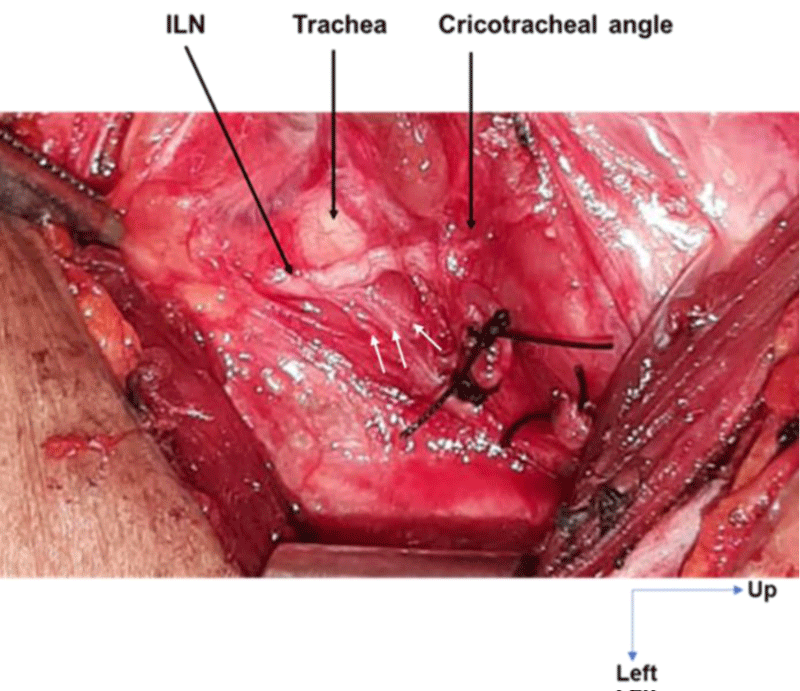
The thickest recurrent nerves were found in brevilinear patients (Figure 3) while the longest recurrent nerves were found in longilinear patients and those with an intermediate morphotype (Figure 4).
The extreme values of ILN dimensions according to morphotype and side are shown in Table 2.
Extra-laryngeal division
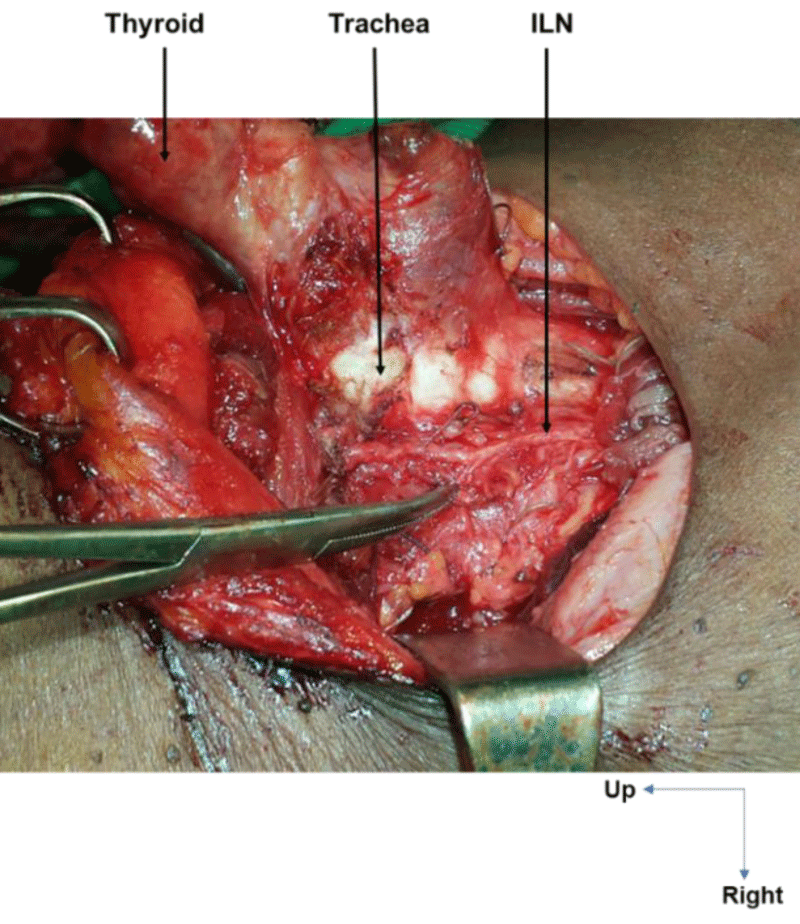
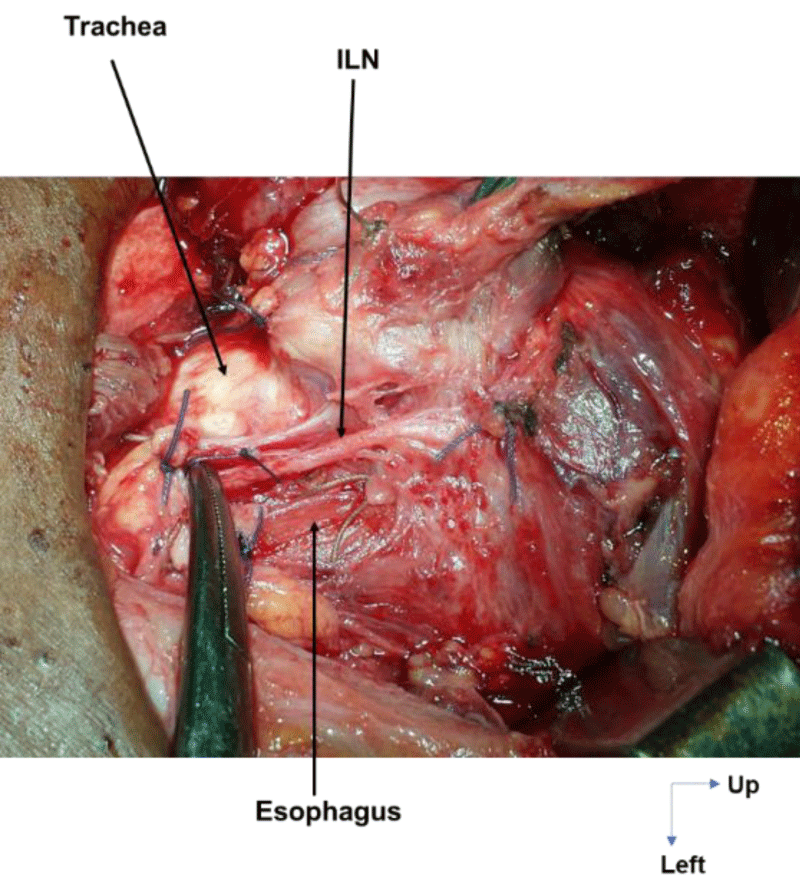
Extra-laryngeal division occurred in all cases at the level of the crossing with the ITA. It was observed in 14 cases (58.3%) on the right and in 25 cases (86.2%) on the left. On the right, the ILN was bifurcated in 10 cases (41.6%, Figure 5), trifurcated in 3 cases (12.5%), and fan-shaped (with 4 branches) in 1 case (4.1%). On the left, it was bifid in 23 cases (79.3%), with an individualized esophageal branch in around 50% of cases, and trifurcated in 2 cases (6.8%, Figure 6).
Level of laryngeal penetration relative to the small horn of the thyroid cartilage
Laryngeal penetration of the recurrent nerve averaged 0.8 cm below the SHTC on the right and 0.7 cm below the SHTC on the left. On both right and left, no ILN had crossed Grüber's ligament before its termination.
Post-operative care
Post-operative management was straightforward. No patient developed postoperative recurrent paresis or paralysis.
Discussion
Methodology
The ILN can be studied during cadaveric dissection or thyroidectomy. Cadaveric dissection offers the advantage of a very detailed study of the recurrent nerve [22-24]. It allows in-depth dissection of the terminal part of the ILN from the cricotracheal angle to its intra-laryngeal distribution, with the aid of tools such as the operating microscope for description and the Vernier for precise measurements [25]. However, it does not reflect the real surgical conditions, notably the distortions induced by thyroid pathology. What's more, it requires fresh cadavers, more technical resources, and legal provisions.
This series corresponds to a live dissection and therefore enables us to appreciate the anatomy of the ILN under real surgical conditions of thyroid pathology [26]. The measuring tools we used, although less precise than the Vernier, are reliable. In fact, the protractor, in which 0 is deposited at the level of the xiphoid process, enables morphotype assessment thanks to a fairly precise measurement of the infrasternal angle or angulus infrasternalis. The suture is a useful tool for assessing ILN dimensions. To measure the length of the ILN, the suture is laid out along the length of the ILN, then the distance of the suture corresponding to the length of the ILN is placed on a ruler to see how it corresponds in cm. The diameter of the ILN is compared with that of a corresponding suture, whose thickness in mm is obtained by placing the suture on the ruler.
lLN route
The course of the recurrent nerve varies according to the side and the distortion induced by thyroid pathology [5]. Often discovered by chance, the non-recurrent ILN remains a delicate situation, increasing the risk of intraoperative recurrent injury [1,7,27].
In this study, no cases of non-recurrent ILN were identified. This reflects the rarity of this anatomical variation in the ILN pathway, found in 0.7% of clinical series and 1.4% of cadaveric series [2].
The ILN pathway is classic in all cases on the right. Variations in the pathway are only observed on the left, and concern 8 nerves (28%). These variations are induced by thyroid pathology, where the recurrent nerve travels on the anterior face of the trachea in 6 cases and on the posterior face of the left lobe of the thyroid gland in the 2 other cases.
Although rarely reported in the literature, ILN path modifications are common and observed in around ¼ of thyroidectomies [13]. Eltokhy [1] in a series of 60 thyroidectomies noted that 12.9% (8 cases) of patients had a variation in the course of the recurrent nerve induced by thyroid pathology.
All these path anomalies represent additional difficulties in dissecting the ILN, thus increasing the risk of recurrent trauma, whose rate varies from 0 to 12% [10,19].
ILN dimensions
The diameter of the recurrent nerve is variable in this series, with a median of 2mm. The mean value of 2mm for ILN diameter is in line with the literature [8,22]. Nevertheless, the extremes of 1mm and 6mm confirm Germain's findings [28,29] on the relative importance of ILN diameter, which can sometimes turn out to be small. The proportion of small ILN noted in this series (16.9% on the right and 3.7% on the left) confirms the greater frequency of smallness on the right, in agreement with the results of Hemmaoui [8] and Page [22], which are 16% and 12.68% on the right and 10% and 9.09% on the left respectively.
This may be partly explained by the longer course of the left recurrent nerve. These fine nerves may favour iatrogenic trauma, as they are difficult to locate and dissect [22].
A study of the correlation of ILN dimensions according to morphotype seemed useful to us and enabled us to observe the presence of the longest recurrent nerves in patients with longilinear and intermediate morphotypes, whereas the thickest or widest recurrent nerves were found in patients with brevilinear morphotypes. However, the path and mode of termination of the ILN are not influenced by morphotype. This correlation of ILN dimensions according to morphotype, which we have not found in the literature, may predicate preoperatively an ease of locating the recurrent nerve in brevilinears, but also its long dissection pathway in longilinears and intermediates, with a greater risk of iatrogenic trauma.
Extra-laryngeal division of the ILN
In this series bifurcation is the most frequent mode of division, observed in 41.6% on the right and 79.3% on the left. Trifurcation and multiple branches are less frequent; 16.6% on the right and 6.8% on the left. Extra-laryngeal divisions of the recurrent nerve are estimated at between 20 and 78% [7,10,16]. The frequency, level, location, and number of branches vary from series to series.
They are more frequent in cadaveric studies, which allow deeper dissection [30]. In a meta-analysis involving 28,387 recurrent nerves, Henry [31] found extra-laryngeal division in approximately 39.2% of intraoperative dissections and 73.3% of cadaveric dissections [31]. Page's study of 251 thyroidectomies involving 205 right and 198 left ILNs found recurrent nerve division in 23.41% on the right and 15.15% on the left [22].
Eltokhy [1], after the dissection of 100 ILNs, noted 10% extra-laryngeal divisions and explained this low rate by the fact that this parameter is not systematically sought in their study.
According to Kim [13], these differences in frequency can be explained by the depth of dissection in the inferior pharyngeal constrictor muscle, by the inclusion of Galen's loop and/or esophageal or tracheal branches as dividing branches, by the use of accessories that simplify branch identification, such as magnifying glasses or endoscopes, and intraoperative monitoring [13].
In this series, division of the recurrent nerve was more frequent on the left, with 25 cases (86.2%) versus 14 cases (58.3%) on the right. This left-hand predominance of ILN division contrasts with that of some authors who have found a right-hand predominance [4,22]. Beneragama [32], in a study of 114 right and 99 left recurrent nerves, found a predominance of right-sided (43%) versus left-sided (28%) division.
These observations allow us to say that the division of the ILN is not influenced by the side. This is confirmed by Henry's meta-analysis [31], which found no significant difference by side.
The number of ILN dividing branches found in this study agrees with that noted by Tang [10] in a cadaveric dissection of 160 ILN, i.e. 2 to 4 branches.
These observations corroborate those of Cernea [4], who in a series of 2154 NLI found 1390 extra-laryngeal divisions with a documented number of branches in 1117 cases, revealing a bifid recurrent nerve in 87.9% of cases, trifurcated in 11.9% of cases and with 4 branches in 0.2% of cases. The left predominance of the division branches is like in our study observed by Eltokhy [1].
A more frequent division of the ILN into two branches has been highlighted in the literature [1,13,16,31,33].
Classically, the recurrent nerve gives rise to two terminal laryngeal branches: an anterior and a posterior branch [1,22]. The majority of authors agree that the posterior, mucosal branch is sensitive and the anterior, muscular branch is motor [1,13,22,31].
With regard to the level of division, according to Steinberg [34], contrary to conventional wisdom, division of the recurrent nerve occurs before it enters the larynx, i.e. in the lower third in 2%, in the middle third in 6% and in the upper third in 92%, most often around 2cm below the crico-thyroid junction. In this study, it occurred in all cases at the level of the crossing with the ITA i.e. close to the upper third of the ILN. This confirms the rarity of division before the crossing with the ITA [7,14,35,18], and confirms the greater vulnerability of the recurrent nerve at the level of its upper segment, due to the frequency of extra-laryngeal division branches, but also to its fixity at this level. This exposes it to trauma through excessive traction on the thyroid gland [2,7].
The extra-laryngeal division of the recurrent nerve represents an anatomical trap, with the risk of confusing a division branch with the nerve trunk [36-39]. This increases the risk of trauma to unrecognized and unidentified branches. Indeed, the risk of iatrogenic injury to the ILN is estimated at 5.2% in the case of extra-laryngeal division, compared with 1.6% in the absence of extra-laryngeal division [13,40]. These lesions may be mechanical, thermal, and/or vascular [19,41].
The absence of recurrent lesions in this series, despite the frequency of extra-laryngeal divisions of the ILN, can be explained in part by the lateral approach, which enabled systematic identification of the trunk of the recurrent, especially when the nerve segment initially discovered was small, and by its meticulous dissection in search of division branches, between its crossing with the ITA and its entry into the crico-tracheal angle.
ILN termination
In this study, the ILN terminated identically on both sides, averaging between 0.7 and 0.8 cm below the SHTC. The termination of the ILN relative to the SHTC is consistent with the literature, which projects it 0.8cm +/- 4 mm below and behind the SHTC, which is easily palpable [1,19]. The laryngeal penetration point represents the most constant landmark of the recurrent nerve, uninfluenced by thyroid pathology or non-recurrence of ILN [1,2,8,19].
Limitations of the study
The limitations of this study are the relatively small sample size and the absence of Intraoperative Neural Monitoring (IONM) for locating the ILN. A larger sample would have enabled a better analysis of morphometric variations of the ILN and those in its course and mode of termination, particularly induced by thyroid pathology. By making it easier to locate the ILN, the detection rate of extralaryngeal nerve branching the IONM reduces operating time and can predict neural injury.
Novel contributions of this study to knowledge
The search for the ILN, its control along its entire length as well as that of all its dividing branches is currently recommended in thyroid surgery. The correlation of ILN dimensions according to morphotype made in this study may predicate preoperatively an ease of locating the recurrent nerve in brevilinears, but also its long dissection pathway in longilinears and intermediates, with a greater risk of iatrogenic trauma. In this series, variations in the pathway only observed on the left and the most frequent division of the recurrent nerve on the left lead us to consider the left thyroid lobe approach more likely to induce recurrent trauma than that of the right thyroid lobe.
The authors recommend systematic measurement of the infrasternal angle before thyroidectomy, as well as a search for anomalies in the path and extralaryngeal dividing branches of the ILN, especially on the left.
Conclusion
Some morphologic anatomical factors were identified in this study as being at risk of recurrential trauma during thyroidectomy, due to the difficulties in locating and/or dissecting the ILN. These include longilinear and intermediate morphotypes, where the longest recurrent nerves were found; abnormalities of the ILN pathway, especially those induced by thyroid pathology, which was only noted on the left; the frequency of spindly recurrent nerves, especially on the right; the presence of extra-laryngeal dividing branches, a frequent situation not influenced by the side being close to the upper third of the ILN, notably at the level of the crossing with the ITA. In order to minimize morbidity, it is essential to search for, monitor, and preserve the integrity of the recurrent nerve and all its dividing branches during thyroid surgery. This involves meticulous dissection right up to its penetration into the cricotracheal angle. The "danger of seeing it" long put forward to justify non-dissection is no longer accepted, hence the need for indirect laryngoscopy or nasofibroscopy before and after thyroidectomy. The laryngeal penetration point represents the most constant landmark of the recurrent nerve, and identification of the ITA can facilitate the identification of the ILN without guaranteeing its preservation.
- Eltokhy AAA. Tips and tricks to save recurrent laryngeal nerve during thyroid surgery. AAMJ. 2015; 13 (4): 83-8
- Uludağ M, Tanal M, İşgör A. A Review of Methods for the Preservation of Laryngeal Nerves During Thyroidectomy. Sisli Etfal Hastan Tip Bul. 2018 Jun 18;52(2):79-91. doi: 10.14744/SEMB.2018.37928. PMID: 32595378; PMCID: PMC7315061.
- Labuschagne JJ, Hammer N. Intra-Operative Detection of a Left-Sided Non-Recurrent Laryngeal Nerve during Vagus Nerve Stimulator Implantation. Medicina (Kaunas). 2020 Sep 23;56(10):489. doi: 10.3390/medicina56100489. PMID: 32977517; PMCID: PMC7598250.
- Cernea CR, Hojaij FC, De Carlucci D Jr, Gotoda R, Plopper C, Vanderlei F, Brandão LG. Recurrent laryngeal nerve: a plexus rather than a nerve? Arch Otolaryngol Head Neck Surg. 2009 Nov;135(11):1098-102. doi: 10.1001/archoto.2009.151. PMID: 19917921.
- Valenzuela-Fuenzalida JJ, Baeza-Garrido V, Navia-Ramírez MF, Cariseo-Ávila C, Bruna-Mejías A, Becerra-Farfan Á, Lopez E, Orellana Donoso M, Loyola-Sepulveda W. Systematic Review and Meta-Analysis: Recurrent Laryngeal Nerve Variants and Their Implication in Surgery and Neck Pathologies, Using the Anatomical Quality Assurance (AQUA) Checklist. Life (Basel). 2023 Apr 24;13(5):1077. doi: 10.3390/life13051077. PMID: 37240722; PMCID: PMC10221453.
- Brasnu D, Ayache D, Hans S, Hartl D, Papon JF. ENT treatise. Medicine-Sciences Flammarion; 2008: 309-13.
- Ardito G, Revelli L, D'Alatri L, Lerro V, Guidi ML, Ardito F. Revisited anatomy of the recurrent laryngeal nerves. Am J Surg. 2004 Feb;187(2):249-53. doi: 10.1016/j.amjsurg.2003.11.001. PMID: 14769313.
- Hemmaoui B, Bouaiti EA, Sahli M, Errami N, Moumni M, Benchafai I, Jahidi A, Zalagh M, Benariba F. Le nerf laryngé inférieur: considérations anatomiques et chirurgicales à propos de 60 thyroïdectomies [The inferior laryngeal nerve: anatomical and surgical considerations about 60 thyroidectomy]. Pan Afr Med J. 2019 May 27; 33:58. French. doi: 10.11604/pamj.2019.33.58.14271. PMID: 31448020; PMCID: PMC6689848.
- Hillermann CL, Tarpey J, Phillips DE. Laryngeal nerve identification during thyroid surgery -- feasibility of a novel approach. Can J Anaesth. 2003 Feb;50(2):189-92. doi: 10.1007/BF03017855. PMID: 12560313.
- Tang WJ, Sun SQ, Wang XL, Sun YX, Huang HX. An applied anatomical study on the recurrent laryngeal nerve and inferior thyroid artery. Surg Radiol Anat. 2012 May;34(4):325-32. doi: 10.1007/s00276-011-0905-8. Epub 2011 Nov 29. PMID: 22124577.
- Gunn A, Oyekunle T, Stang M, Kazaure H, Scheri R. Recurrent Laryngeal Nerve Injury After Thyroid Surgery: An Analysis of 11,370 Patients. J Surg Res. 2020 Nov; 255:42-49. doi: 10.1016/j.jss.2020.05.017. Epub 2020 Jun 13. PMID: 32540579.
- Cho I, Jo MG, Choi SW, Jang JY, Wang SG, Cha W. Some posterior branches of extralaryngeal recurrent laryngeal nerves have motor fibers. Laryngoscope. 2017 Nov;127(11):2678-2685. doi: 10.1002/lary.26595. Epub 2017 Apr 20. PMID: 28425617.
- Kim HY, Liu X, Sun H, Chai YJ, Chung WY, Dionigi G. Monitoring the Bifurcated Recurrent Laryngeal Nerve. J Endocr Surg. 2017;17(2) :31-41
- Noussios G, Chatzis I, Konstantinidis S, Filo E, Spyrou A, Karavasilis G, Katsourakis A. The Anatomical Relationship of Inferior Thyroid Artery and Recurrent Laryngeal Nerve: A Review of the Literature and Its Clinical Importance. J Clin Med Res. 2020 Oct;12(10):640-646. doi: 10.14740/jocmr4296. Epub 2020 Sep 21. PMID: 33029270; PMCID: PMC7524559.
- Pandey AK, Varma A, Bansal C, Bhardwaj A. An Exposition on Surgical Experiences in Identification, Exposure, and Injuries of Recurrent Laryngeal Nerve (RLN) During Thyroid Operations: Gleanings, Narrative, and the Reflections. Indian J Otolaryngol Head Neck Surg. 2023 Sep;75(3):1363-1369. doi: 10.1007/s12070-023-03541-7. Epub 2023 Feb 14. PMID: 37636600; PMCID: PMC10447788.
- Lubrano D, Levy-Chazal N, Araya Y, Avisse C. La recherche du nerf laryngé inférieur ou récurrent lors d'une lobectomie thyroïdienne [Identification of recurrent inferior laryngeal nerve during a thyroid lobectomy]. Ann Chir. 2002 Jan;127(1):68-72. French. doi: 10.1016/s0003-3944(01)00674-5. PMID: 11833312.
- Shen C, Xiang M, Wu H, Ma Y, Chen L, Cheng L. Routine exposure of recurrent laryngeal nerve in thyroid surgery can prevent nerve injury. Neural Regen Res. 2013 Jun 15;8(17):1568-75. doi: 10.3969/j.issn.1673-5374.2013.17.004. PMID: 25206452; PMCID: PMC4145966.
- Kostek M, Caliskan O, Yanar C, Cakir Y, Uludag M. The Most Common Anatomical Variation of Recurrent Laryngeal Nerve: Extralaryngeal Branching. Sisli Etfal Hastan Tip Bul. 2021 Sep 24;55(3):294-303. doi: 10.14744/SEMB.2021.93609. PMID: 34712069; PMCID: PMC8526224.
- Goldenberg D, Randolph GW. Intraoperative Complications: The ‘Classic’ issues (the recurrent laryngeal nerve (PART III) in thyroid surgery: preventing and managing complications, first edition. John Wiley & Sons, Ltd: 2013; 119-27.
- Kaplan EL, Salti GI, Roncella M, Fulton N, Kadowaki M. History of the recurrent laryngeal nerve: from Galen to Lahey. World J Surg. 2009 Mar;33(3):386-93. doi: 10.1007/s00268-008-9798-z. PMID: 19023621.
- Koumare AK, Ongoiba N, Sissoko F, Berete S, Diop AT, Sidibe Y, et al. Inferior laryngeal nerve: anatomy and operative lesions. E-memoires of the French national academy of surgery. 2002; 1 (2): 8-11
- Page C, Foulon P, Strunski V. The inferior laryngeal nerve: surgical and anatomic considerations. Report of 251 thyroidectomies. Surg Radiol Anat. 2003 Jul-Aug;25(3-4):188-91. doi: 10.1007/s00276-003-0129-7. Epub 2003 Aug 9. PMID: 12910381.
- Ngo Nyeki AR, Njock LR, Miloundja J, Evehe Vokwely JE, Bengono G. Recurrent laryngeal nerve landmarks during thyroidectomy. Eur Ann Otorhinolaryngol Head Neck Dis. 2015 Nov;132(5):265-9. doi: 10.1016/j.anorl.2015.08.002. Epub 2015 Aug 31. PMID: 26338514.
- Liddy W, Wu CW, Dionigi G, Donatini G, Giles Senyurek Y, Kamani D, Iwata A, Wang B, Okose O, Cheung A, Saito Y, Casella C, Aygun N, Uludag M, Brauckhoff K, Carnaille B, Tunca F, Barczyński M, Kim HY, Favero E, Innaro N, Vamvakidis K, Serpell J, Romanchishen AF, Takami H, Chiang FY, Schneider R, Dralle H, Shin JJ, Abdelhamid Ahmed AH, Randolph GW. Varied Recurrent Laryngeal Nerve Course Is Associated with Increased Risk of Nerve Dysfunction During Thyroidectomy: Results of the Surgical Anatomy of the Recurrent Laryngeal Nerve in Thyroid Surgery Study, an International Multicenter Prospective Anatomic and Electrophysiologic Study of 1000 Monitored Nerves at Risk from the International Neural Monitoring Study Group. Thyroid. 2021 Nov;31(11):1730-1740. doi: 10.1089/thy.2021.0155. PMID: 34541890.
- Sepulveda A, Sastre N, Chousleb A. Topographic anatomy of the recurrent laryngeal nerve. J Reconstr Microsurg. 1996 Jan;12(1):5-10. doi: 10.1055/s-2007-1006445. PMID: 8618227.
- Pradeep PV, Jayashree B, Harshita SS. A Closer Look at Laryngeal Nerves during Thyroid Surgery: A Descriptive Study of 584 Nerves. Anat Res Int. 2012; 2012:490390. doi: 10.1155/2012/490390. Epub 2012 Jun 12. PMID: 22737584; PMCID: PMC3378964.
- Gurleyik E. Three variations of the laryngeal nerve in the same patient: a case report. J Med Case Rep. 2011 Jul 1; 5:266. doi: 10.1186/1752-1947-5-266. PMID: 21722360; PMCID: PMC3141713.
- Germain MA, Trotoux J, Bruneau X. Les nerfs laryngés. Etude histomorphométrique et cartographie [Laryngeal nerves. A histomorphometric and cartographic study]. Ann Otolaryngol Chir Cervicofac. 1986;103(6):379-88. French. PMID: 3538976.
- Ghosh A, Chaudhury S. Cadaveric Measurements of the Left Recurrent Laryngeal Nerve, Ligamentum Arteriosum, Aortic Arch, and Pulmonary Artery in the Thorax with Clinical Implications and Comparison Between Two Sexes in the American Population. Cureus. 2019 Jun 4;11(6):e4828. doi: 10.7759/cureus.4828. PMID: 31403016; PMCID: PMC6682338.
- Schweizer V, Dörfl J. The anatomy of the inferior laryngeal nerve. Clin Otolaryngol Allied Sci. 1997 Aug;22(4):362-9. doi: 10.1046/j.1365-2273.1997.00028.x. PMID: 9298614.
- Henry BM, Vikse J, Graves MJ, Sanna S, Sanna B, Tomaszewska IM, Tubbs RS, Tomaszewski KA. Extralaryngeal branching of the recurrent laryngeal nerve: a meta-analysis of 28,387 nerves. Langenbecks Arch Surg. 2016 Nov;401(7):913-923. doi: 10.1007/s00423-016-1455-7. Epub 2016 Jun 2. PMID: 27251487; PMCID: PMC5086344.
- Beneragama T, Serpell JW. Extralaryngeal bifurcation of the recurrent laryngeal nerve: a common variation. ANZ J Surg. 2006 Oct;76(10):928-31. doi: 10.1111/j.1445-2197.2006.03899.x. PMID: 17007624.
- Yin C, Song B, Wang X. Anatomical Variations in Recurrent Laryngeal Nerves in Thyroid Surgery. Ear Nose Throat J. 2021 Dec;100(10_suppl):930S-936S. doi: 10.1177/0145561320927565. Epub 2020 Jun 4. PMID: 32493053.
- Steinberg JL, Khane GJ, Fernandes CM, Nel JP. Anatomy of the recurrent laryngeal nerve: a redescription. J Laryngol Otol. 1986 Aug;100(8):919-27. doi: 10.1017/s0022215100100325. PMID: 3746108.
- Darr EA, Randolph GW. Management of laryngeal nerves and parathyroid glands at thyroidectomy. Oral Oncol. 2013 Jul;49(7):665-70. doi: 10.1016/j.oraloncology.2013.03.438. Epub 2013 Apr 16. PMID: 23597767.
- Saira N, Adnan YM, Waseem K, Khursheed A, Fazal R. Branching patterns of recurrent laryngeal nerve in thyroidectomy patients. J Saidu Med Coll Swat. 2022 Sep; 12(3):143-7.
- Gaurav K, Roy D, Rajak R, Hashmi A, Khan ZM. Anatomical variation of recurrent laryngeal nerve encountered during thyroid surgeries in tribal population of Jharkhand, a prospective analysis. SJHR-Africa. 4(9):6. https://doi.org/10.51168/sjhrafrica.v4i9.652
- Krishnan PB, Santosh MP. An atypical bilateral trifurcation of recurrent laryngeal nerve. BMC Surg. 2022 May 13;22(1):176. doi: 10.1186/s12893-022-01624-w. PMID: 35562686; PMCID: PMC9101918.
- Thomas AM, Fahim DK, Gemechu JM. Anatomical Variations of the Recurrent Laryngeal Nerve and Implications for Injury Prevention during Surgical Procedures of the Neck. Diagnostics (Basel). 2020 Sep 4;10(9):670. doi: 10.3390/diagnostics10090670. PMID: 32899604; PMCID: PMC7555279.
- Piperos T, Kaklamanos I, Chrysikos D, Zarokosta M, Boumpa E, Zoulamoglou M, Kalles V, Gkogka GI, Mariolis-Sapsakos T. Abnormal distance of the extralaryngeal bifurcation point of the recurrent laryngeal nerve from the cricothyroid joint. J Surg Case Rep. 2018 Jan 17;2018(1):rjx257. doi: 10.1093/jscr/rjx257. PMID: 29383239; PMCID: PMC5786220.
- Zhao ZL, Wei Y, Peng LL, Li Y, Lu NC, Yu MA. Recurrent Laryngeal Nerve Injury in Thermal Ablation of Thyroid Nodules-Risk Factors and Cause Analysis. J Clin Endocrinol Metab. 2022 Jun 16;107(7):e2930-e2937. doi: 10.1210/clinem/dgac177. PMID: 35311971.
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
